 The n values and the electronic configurations (e.c. Where N is the Avogadro's number (6.022x10 -23), and n is a number related to the electronic configurations of the ions involved. Lattice energy refers to the energy which is released while two oppositely charged gaseous ions attract to each other and form an ionic solid. Which chemical process is associated with the lattice energy for sodium chloride Nack (s) Na (8) + CI (8) NaCl (s) - Na (8) + Cl (8) Na (s) + 1/2 Cl2 (8) -NaCl (s) NaCl (s) + H20 (1) - Nat (aq) + Cl (aq) This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. This expression takes into account both the Born interactions as well as the Coulomb attractions. The standard is named ASTM E534-13 and is the standard test methods for chemical analysis of sodium chloride. 8.24Does the lattice energy of an ionic solid increase or decrease (i) as the chargesof the ions increase, (ii) as the sizes of the ions increase Arrange the. 
(Given: Lattice energy of NaCl - 777.8kJ mol-1 ), hydration energy - 774.1kJ mol-1 and S 0.043KJK-1 mol-1 at 298K. An estimate of the strength of the bonds in an ionic compound can be obtained by measuring the lattice energy of the compound, which is the energy given off when oppositely charged ions in the gas phase come together to form a solid. The Born-Landé equation is a concept originally formulated in 1918 by the scientists Born and Landé and is used to calculate the lattice energy (measure of the strength of bonds) of a compound. Standard Sodium chloride has an international standard that is created by ASTM International. That means that we will have to use theoretical values of their lattice enthalpies.\] Click hereto get an answer to your question Calculate the free energy change when 1 mole of NaCl is dissolved in water at 298K. Writing again the lattice energy as the sum of the Madelung energy and of the Born repulsive energy in the form. The equation for the lattice energy is the reverse of the equation in Step 5 in the figure below, for the formation of the solid from its ions which releases 787 kJ/mol of energy. The process absorbs energy, and is highly endothermic. Data: Enthalpy of sublimation of Na(s) 107.5 kJ/mol 1st ionization energy of Na(g) 496 kJ/mol Bond dissociation energy of Cl2(g) 242.58 kJ/mol 1st electron affinity of Cl(g) 348.57 kJ/mol Standard enthalpy of formation of NaCl(s) 411. The lattice energy for NaCl is: NaCl (s) Na + (g) + Cl (g) H lattice +787 kJ/mol. Let's look at this in terms of Born-Haber cycles of and contrast the enthalpy change of formation for the imaginary compounds MgCl and MgCl 3. 1) Calculate the lattice energy of NaCl(s) using a BornHaber cycle. (b) In terms of atomic structure explain why the first-ionization energy is. It turns out that MgCl 2 is the formula of the compound which has the most negative enthalpy change of formation - in other words, it is the most stable one relative to the elements magnesium and chlorine. comUnit 5: In AP Classroom Complete Personal Progress Check Unit 8: In AP Ap. Theoretical calculation of lattice energy : Sodium Chloride, NaCl, Na+ 6 : Cl- 6 Cesium Chloride, CsCl, Cs+ 8 : Cl- 8 Zinc Blende, ZnS, Zn2+ 4 : S2. It is best thought of as a face-centered cubic array of anions with an interpenetrating fcc cation lattice (or vice-versa).  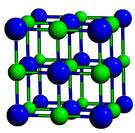
The question arises as to why, from an energetics point of view, magnesium chloride is MgCl 2 rather than MgCl or MgCl 3 (or any other formula you might like to choose). Solution Verified by Toppr Correct option is A) The energy released during the formation of a solid ionic compound from its constituent gaseous ions or in other words the energy used to decompose the ionic solid into its constituent cation and anion is called lattice energy. The Sodium Chloride Structure General Notes NaCl has a cubic unit cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
